[ad_1]
Surgeons at Duke University successfully implanted a next-generation artificial heart in a man with heart failure, a first for any hospital in North America.
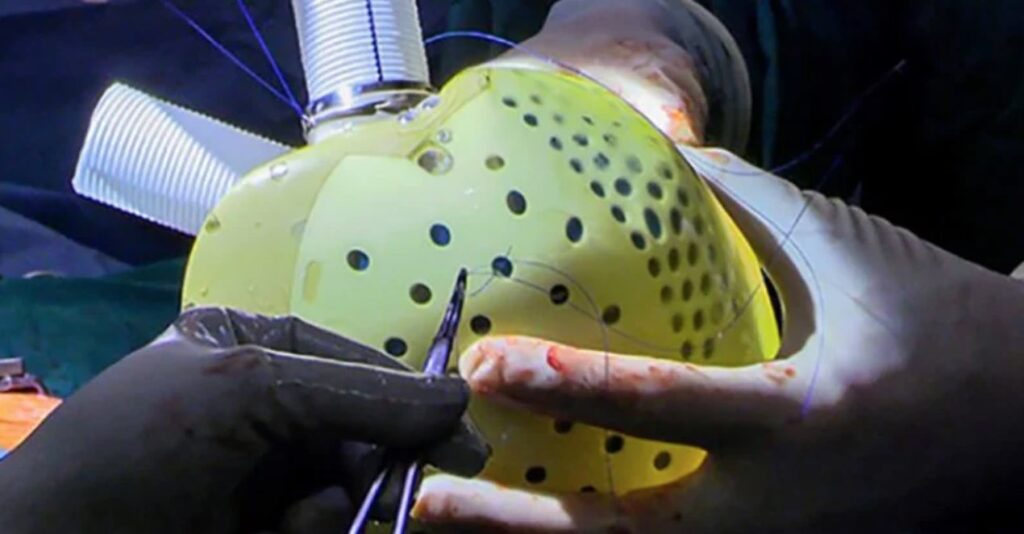
The artificial heart was developed by a French company, CARMAT, and has been approved for use and sale in Europe.
Last year, the company received approval from the US FDA to begin studies and enroll 10 patients with end-stage biventricular heart failure (people who are on the waiting list for a heart donor) and offer a bridge save their lives before transplant.
“We are encouraged that our patient is doing so well after Monday’s procedure,” said Dr. Carmelo Milano, transplant surgeon and principal investigator of the device study at Duke. “As we evaluate this device, we are excited and hopeful that patients who otherwise have little or no choice may have a lifeline.”
RELATED: Stem cell-based ‘cure’ for type 1 diabetes is coming, with the launch of FDA trials
North Carolina patient Matthew Moore is just 39 years old and was referred to Duke in June after a sudden and unexpected diagnosis of heart failure. Moore and his wife, Rachel, recently adopted their two-year-old adopted son, Marshall, and came to Duke expecting only to have heart bypass surgery.
However, as Moore’s condition rapidly deteriorated, traditional options, including transplantation, became too risky. Meanwhile, Duke was one of three transplant centers in the United States selected to join the study of the device, and the procedure team received specialized training to prepare for implant surgery.
Matthew’s wife, a nurse, said: “Both Matthew and I are so grateful that we have been given the opportunity to participate in something that has the potential to impact so many lives. We are simply taking it day by day and we hope that everything continues to progress well ”.
The artificial heart device called ‘Aeson’, is an implantable prosthesis that includes biological valves derived from bovine tissue and works with an external power source.
CHECK: Student builds a life-saving device that can instantly stop bleeding from stab wounds
If the device receives FDA approval, it would bring hope to transplant patients whose hearts require help pumping blood through both chambers. Current technology, particularly a left ventricular assist device (LVAD), supports a single chamber.
“Due to the shortage of donor hearts, many patients die while waiting for a heart transplant,” said Schroder, a transplant surgeon who led the implant procedure. “We are hopeful for new options to help these patients, many like Mr. Moore, who have a devastating disease and cannot otherwise be considered for transplantation.”
CLOCK a CBS report …
MORE: The venom of an extremely poisonous caterpillar may contain a life-saving healing tonic
SHARE the heartwarming innovation on social media …
[ad_2]
Source link here