[ad_1]
Scientists have discovered how to modify the incomparable CRISPR gene-editing tool to extend its reach to the epigenome, which controls how genes are turned on or off.
Researchers at the University of California, San Francisco and MIT’s nonprofit Whitehead Institute for Biomedical Research have already used the tool in the laboratory to primarily turn off the gene that produces the Tau protein, which has been implicated in the Alzheimer disease.
The novel CRISPR-based tool called “CRISPRoff” allows scientists to turn off almost any gene in human cells without doing a single edit of the genetic code, and once a gene is turned off, it remains inert in the cell’s descendants for hundreds of generations. , unless it is turned back on with a companion tool called CRISPRon.
Because the epigenome plays a central role in many diseases, from viral infection to cancer, CRISPRoff technology may one day lead to powerful epigenetic therapies that are safer than conventional CRISPR therapies because it does not involve any DNA editing.
“Although gene and cell therapies are the future of medicine, there are potential safety concerns around permanent genome change, so we are trying to find other ways to use CRISPR to treat disease,” said Luke Gilbert, PhD, UCSF professor and co-lead author of the new paper, published April 9 daily Cell.
How it was built
Conventional CRISPR is equipped with two pieces of molecular hardware that make it an effective gene editing tool. One component is a DNA cutting enzyme, which gives CRISPR the ability to alter DNA sequences. The other is a locating device that can be programmed to focus on any DNA sequence of interest, imparting precise control over where edits are made.
To build CRISPRoff, the researchers dispensed with the conventional CRISPR DNA-cutting enzymatic function, but retained the localization device, creating a shortened CRISPR capable of targeting any gene. Then they tied an enzyme to this barebones CRISPR. But instead of splicing DNA, this enzyme acts on the epigenome, which consists of proteins and small molecules that attach to DNA and control when and where genes are turned on or off.
The new tool targets a particular epigenetic feature known as DNA methylation, which is one of the many molecular parts of the epigenome. When DNA is methylated, a small chemical tag known as a methyl group attaches to DNA, silencing nearby genes. Although DNA methylation occurs naturally in all mammalian cells, CRISPRoff offers scientists unprecedented control over this process.
Another tool described in the paper, called CRISPRon, removes the methylation marks deposited by CRISPRoff, making the process completely reversible.
“We now have a simple tool that can silence the vast majority of genes,” said Jonathan Weissman, PhD, fellow at the Whitehead Institute, a co-lead author of the new paper, and a former UCSF faculty member. “We can do this for multiple genes at the same time without damaging the DNA, and in a way that can be reversed. It’s a great tool for controlling gene expression. “
The ‘big surprise’ changes a basic principle
Building on earlier work by a group in Italy, the researchers were confident that CRISPRoff would be able to silence specific genes, but they suspected that around 30 percent of human genes would not respond to the new tool.
DNA consists of four genetic letters: A, C, G, T, but generally only C’s next to G’s can be methylated. To complicate matters, scientists have long believed that methylation could only silence genes at sites in the genome where CG sequences are highly concentrated, regions known as “CpG islands.”
Since nearly a third of human genes lack CpG islands, the researchers assumed that methylation would not turn off these genes. But his CRISPRoff experiments overturned this epigenetic dogma.
“What was thought before this work was that 30 percent of genes that do not have CpG islands were not controlled by DNA methylation.” Gilbert said. “But our work clearly shows that you don’t need a CpG island to turn off genes by methylation. That, for me, was a big surprise. “
Improvement of the therapeutic potential of CRISPRoff
Easy-to-use epigenetic editors like CRISPRoff have enormous therapeutic potential, in large part because, like the genome, the epigenome can be inherited.
MORE: Revolutionary CRISPR-based genome editing system kills cancer cells ‘permanently’ in the laboratory
When CRISPRoff silences a gene, the gene not only remains inactivated in the treated cell, but also remains inactivated in the descendants of the cell as it divides, for up to 450 generations.
To the researchers’ surprise, this held true even in maturing stem cells. Although the transition from stem cell to differentiated adult cell involves significant rewiring of the epigenome, the methylation marks deposited by CRISPRoff were faithfully inherited in 90 percent of the cells that made this transition, showing that the cells retain a memory of epigenetic modifications made by the CRISPRoff System even when cell type changes.
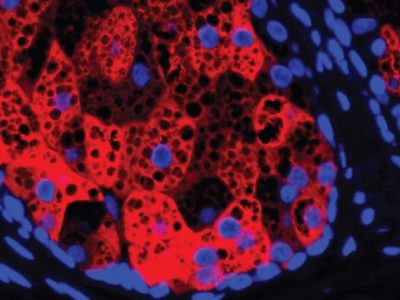
Alzheimer’s and Tau protein
They selected a gene to use as an example of how CRISPRoff could be applied to therapeutics: the gene that encodes the Tau protein, which is implicated in Alzheimer’s disease. After testing the method in neurons, they found that the use of CRISPRoff could be used to reduce the expression of Tau, although not entirely. “What we show is that this is a viable strategy to silence Tau and prevent that protein from being expressed,” says Weissman. “The question is, then, how do you deliver this to an adult? And would it really be enough to impact Alzheimer’s disease? Those are big open questions, especially the last one. “
Even if CRISPRoff does not lead to Alzheimer’s therapies, there are many other conditions it could apply to. Their findings suggest that CRISPRoff would only need to be given once to have long-lasting therapeutic effects, making it a promising approach for treating rare genetic disorders, including Marfan syndrome, which affects connective tissue, Job’s syndrome, a immune system disorder and certain forms of cancer – which are caused by the activity of a single damaged copy of a gene.
WHAT’S MORE: For the first time, scientists have cured living creatures from HIV and completely removed the virus from DNA
While delivery to specific tissues remains a challenge, “we demonstrated that it can be transiently delivered as DNA or as RNA, the same technology that is the foundation of the Moderna and BioNTech coronavirus vaccine,” says Weissman.
Because the epigenome plays a central role in so many diseases, this exciting new technology may one day lead to powerful therapies to tackle our most deadly foes, although “more work is needed to realize its full therapeutic potential.”
EDIT your social media feed with this hopeful good news …
[ad_2]
source material