Are Your Ears Ringing From Tinnitus? This Non-Invasive Nerve Stimulation Could Help Millions
In the largest clinical trial of its kind, researchers have shown that the combination of acoustic and electrical stimulation of the tongue can significantly reduce tinnitus, commonly described as “ringing in the ears.” They also found that the therapeutic effects can be maintained for up to 12 months after treatment.
The findings could help millions of people, as tinnitus affects between 10 and 15 percent of the population worldwide.
The research was recently published as a cover story for the leading peer-reviewed journal. Translational science medicine.
The study represents the largest and longest follow-up clinical trial ever conducted in the field of tinnitus for a medical device with 326 enrolled participants, providing evidence on the safety, efficacy and patient tolerability of bimodal neuromodulation for the treatment of tinnitus .
About 86 percent of participants who complied with treatment reported an improvement in the severity of their tinnitus symptoms when assessed after 12 weeks of treatment, and many experienced sustained benefit 12 months after treatment.
“I am really proud of our company’s ability to conduct a large-scale, randomized clinical trial in two countries,” said University of Minnesota associate professor Hubert. Lim said. “This study tracked post-treatment therapeutic effects for 12 months, which is the first time that the field of tinnitus has evaluated the long-term results of a medical device approach. The results are very exciting and I look forward to continuing our work to develop a bimodal neuromodulation treatment that helps as many tinnitus patients as possible. “
Although the study, which was sponsored by Neuromod Devices, had consistent therapeutic results at both clinical sites, with no serious adverse events.
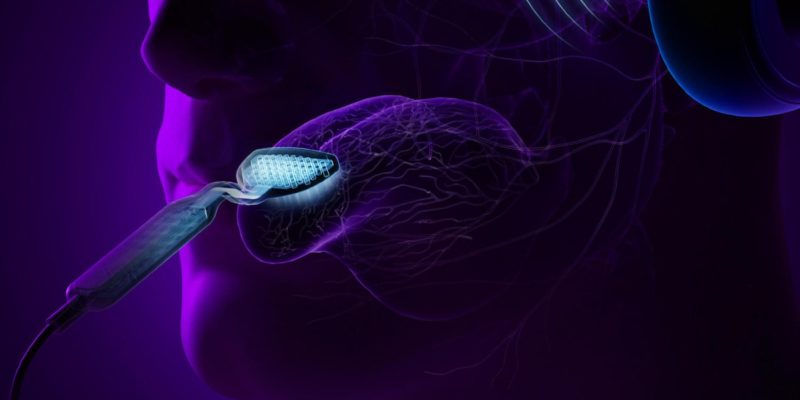
The tinnitus treatment device used in the study, now branded as Lenire®, was developed by Neuromod Devices and consists of wireless (Bluetooth®) headphones that transmit streams of audio tones layered with broadband noise layers to both ears, combined with pulses of electrical stimulation delivered to 32 electrodes on the tip of the tongue by a proprietary device registered as Tonguetip®.
RELATED: An 8-year-old boy sees the stars for the first time after his blindness is treated with gene therapy
The timing, intensity, and delivery of the stimuli are controlled by an easy-to-use handheld controller that each participant is trained to operate.
Before using the treatment for the first time, the device is configured according to the patient’s hearing profile and optimized to the patient’s level of sensitivity for tongue stimulation.
For the test, participants were instructed to wear the Lenire® device for 60 minutes a day for 12 weeks.
Of the 326 participants enrolled, 83.7 percent used the device at or above the 36-hour minimum compliance level during the 12-week treatment period.
For the primary endpoints, participants achieved a statistically and clinically significant reduction in the severity of tinnitus symptoms.
When the treatment was completed, the participants returned their devices and were evaluated at three follow-up visits for up to 12 months.
Just over 66 percent of participants who completed the exit survey stated that they had benefited from using the device, and 77.8 percent said they would recommend treatment for other people with tinnitus.
CHECK OUT: Key ingredient in coronavirus testing comes from Yellowstone’s heated pools
Study participants were assessed and selected according to a predefined list of inclusion and exclusion criteria to ensure that the trial had a widely distributed sample of the tinnitus population.
(CLOCK the video about this revolutionary device below).
(Source: University of Minnesota)
SHARE this good news with friends who need to hear it on social media …